 
The Joint Commission (TJC) is the accrediting body for hospitals. 28 Many poisonings are acute unanticipated emergencies, which would presumably invoke the protections of the Emergency Medical Treatment and Labor Act of 1986, which mandates public access to emergency services regardless of ability to pay. Death and serious harm resulted from the delay to administer appropriate antidotes, reversal agents, and rescue agents. The most common adverse reactions (>5%) include transient chromaturia, erythema, oxalate crystals in urine, rash, increased blood pressure, nausea, headache and infusion site reactions.Sufficient antidote stocking is a best practice failure to do so presents a clear and present danger to patient safety.Breastfeeding is not recommended during treatment with CYANOKIT.In animal studies, hydroxocobalamin administered to pregnant rats and rabbits during the period of organogenesis caused skeletal and soft tissue abnormalities, including alterations in the central nervous system, at exposures similar to human exposures at the therapeutic dose. There are risks to the pregnant woman and fetus associated with untreated cyanide poisoning. Available data from cases reported in the published literature and postmarketing surveillance with CYANOKIT use in pregnant women are insufficient to identify a drug-associated risk for major birth defects, miscarriage, or adverse maternal and fetal outcomes.While it is not known if the skin redness predisposes to photosensitivity, patients should be advised to avoid direct sun while their skin remains discolored. 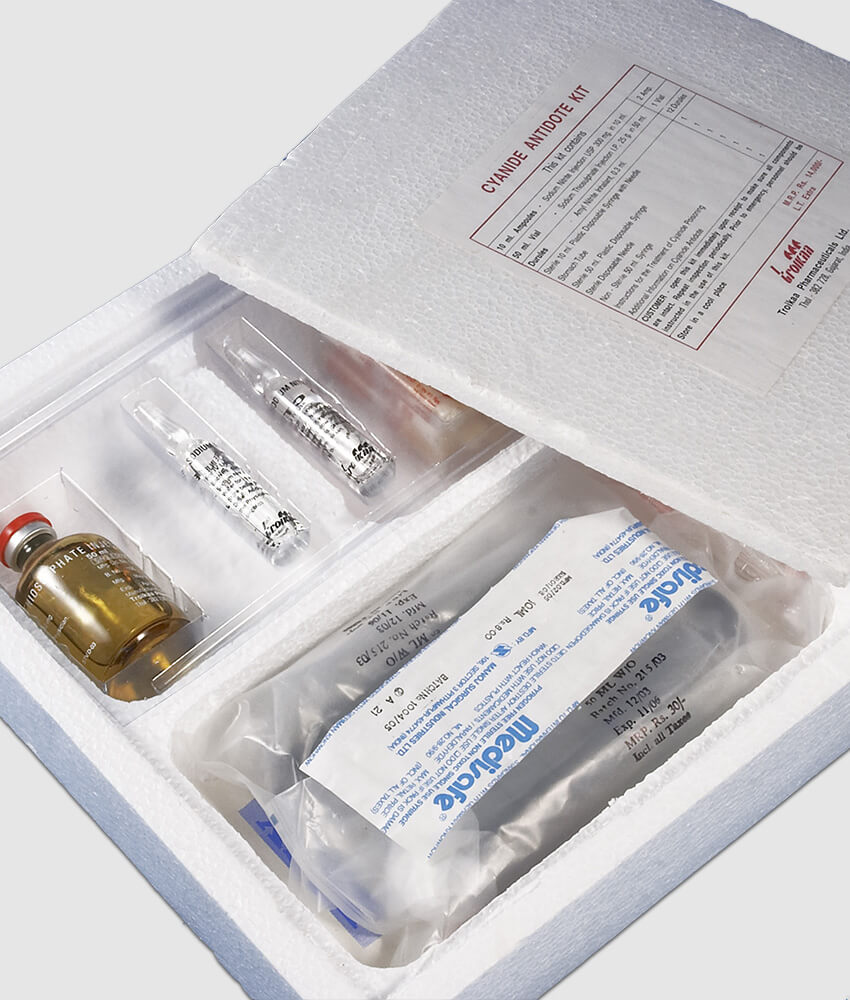
It therefore has potential to cause photosensitivity. Hydroxocobalamin absorbs visible light in the UV spectrum.This should be considered before hemodialysis is initiated in patients treated with hydroxocobalamin. Because of its deep red color, hydroxocobalamin may cause hemodialysis machines to shut down due to an erroneous detection of a “blood leak”.While determination of blood cyanide concentration is not required for management of cyanide poisoning and should not delay treatment with CYANOKIT, collecting a pretreatment blood sample may be useful for documenting cyanide poisoning as sampling post-CYANOKIT use may be inaccurate.Be aware of this when reporting and interpreting laboratory results. Because of its deep red color, hydroxocobalamin has been found to interfere with colorimetric determination of certain laboratory parameters (e.g., clinical chemistry, hematology, coagulation, and urine parameters).Substantial increases in blood pressure may occur following CYANOKIT therapy.Monitor renal function for 7 days following CYANOKIT therapy. Acute renal failure with acute tubular necrosis, renal impairment and urine calcium oxalate crystals have been reported following CYANOKIT therapy.Allergic reactions including angioneurotic edema have also been reported in postmarketing experience. Allergic reactions may include: anaphylaxis, chest tightness, edema, urticaria, pruritus, dyspnea, and rash.Consider alternative therapies, if available, in patients with known anaphylactic reactions to hydroxocobalamin or cyanocobalamin.Risk of Anaphylaxis and Other Hypersensitivity Reactions Consideration should be given to decontamination measures based on the route of exposure. In conjunction with CYANOKIT, treatment of cyanide poisoning must include immediate attention to airway patency, adequacy of oxygenation and hydration, cardiovascular support, and management of seizures. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
